At the Intersection
of Science and IT
Digitizing scientific workflows in regulated manufacturing environments to support data integrity, efficiency, and compliance with Connected Plant™.
Live from Europe CEC 26: LabWare and Phizzle Partner to Eliminate Manual Instrument Data Transfer in Pharmaceutical Cleanroom Labs. Learn More
Digitizing scientific workflows in regulated manufacturing environments to support data integrity, efficiency, and compliance with Connected Plant™.
Eliminate pen and paper processes, multi-step data entry SOPs and clunky middleware solutions to synchronize critical manufacturing data with LIMS, MES, ELN or QMS.
See Our Solution
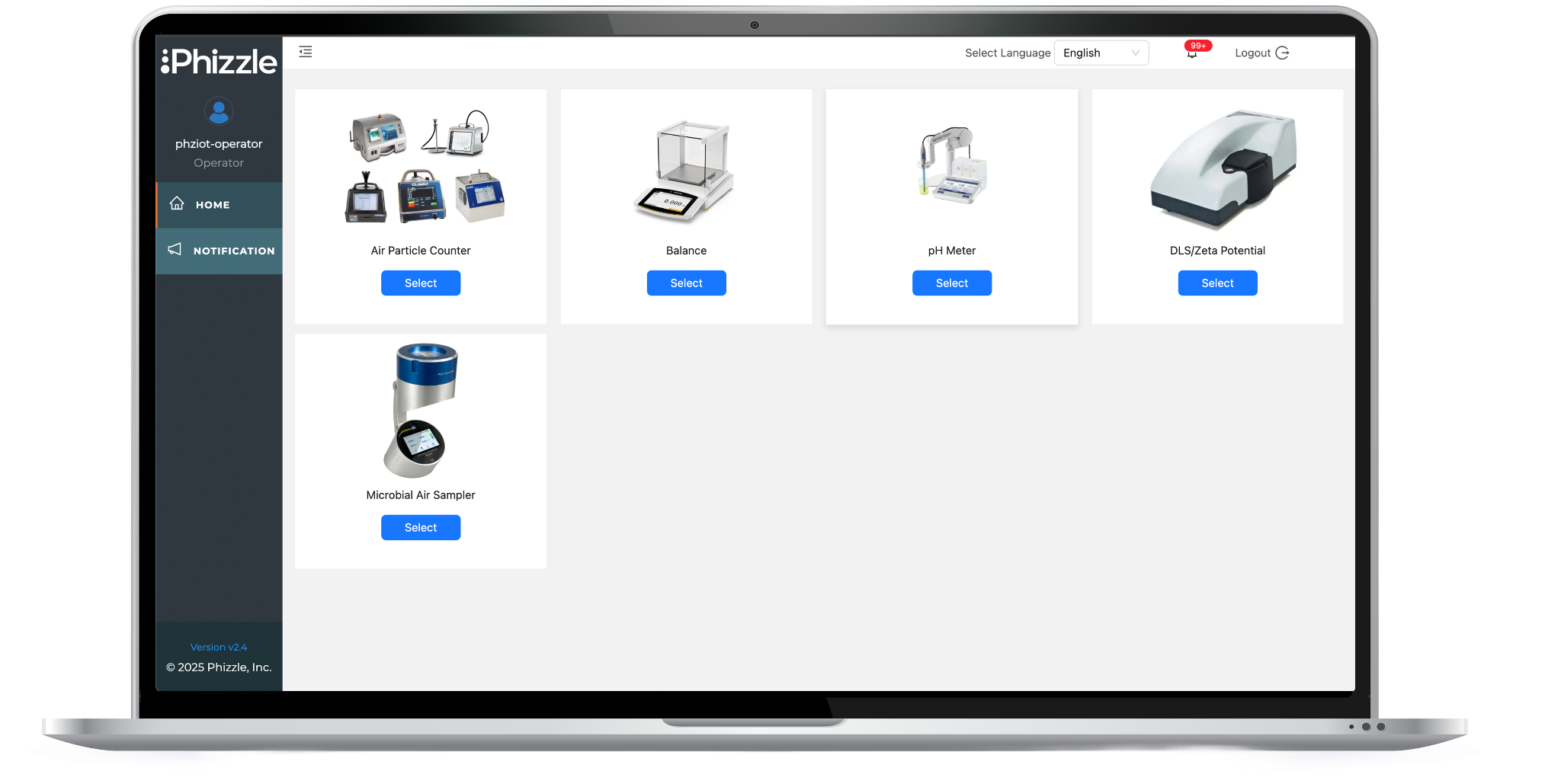
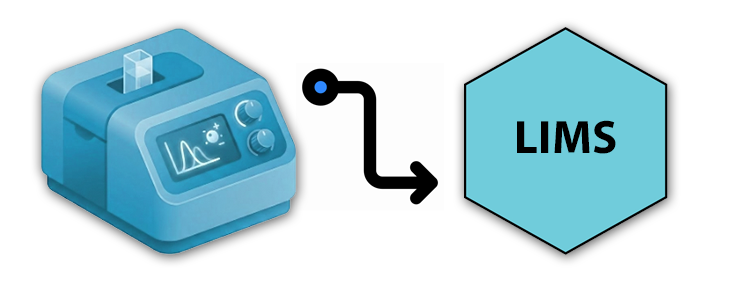
Automate data capture, format standardization and synchronization with LIMS, MES, ELN or QMS. Ensure 21 CFR Part 11 compliance with a built-in review, electronic signature process and detailed audit trails.
Learn MoreEnable hands-free data capture, automated formatting and data synchronization with ERP, MES, or QMS. Support AS9100 and NADCAP compliance requirements with comprehensive traceability and detailed audit trails.
Learn MoreStreamline critical control point monitoring and documentation by synchronizing real-time quality data directly with food safety management systems. Support HACCP and 21 CFR Part 11 compliance requirements with continuous monitoring, electronic record keeping and detailed audit trails.
Learn MoreSee how a Top-3 Pharma is saving $1.5M a year in labor costs and cut failure reports by 80%
View the Case StudySee how Connected Plant™ eliminates manual data entry and integrates directly with your LIMS, MES, ELN, or QMS.
Request a Demo